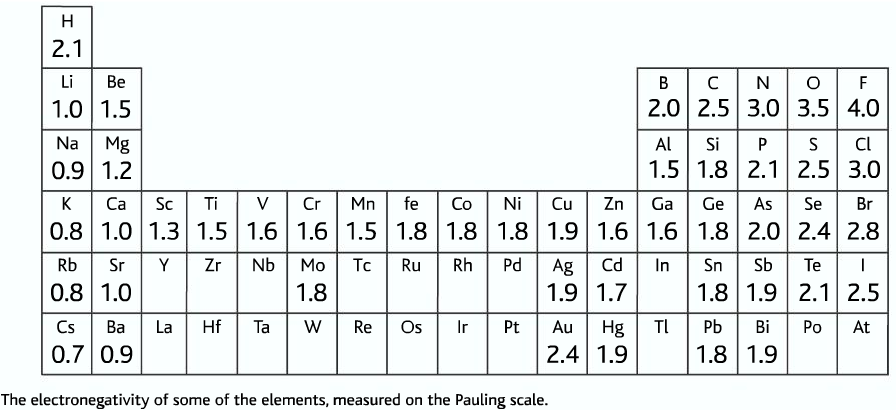

- To list the elements order by electronegativity, click on the table headers. You can print the list of elements by hitting the print button below. The element which has the highest electronegativity value is Fluorine with 3.98 χ. And the element which has the lowest electronegativity value is Francium in 0.7 χ.
- Electronegativity is a chemical property which describes how well an atom can attract an electron to itself. Values for electronegativity run from 0 to 4. Electronegativity is used to predict whether a bond between atoms will be ionic or covalent. It can also be used to predict if the resulting molecule will be polar or nonpolar.
Electronegativity was first measured and developed by Linus Pauling in 1932. He created the Pauling scale, giving each element a value that corresponds to its ability to attract electrons. The elements range in value from 0.7 (caesium and francium), the least electronegative, to 4.0 (fluorine). Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. What happens if two atoms of equal electronegativity bond together? Consider a bond between two atoms, A and B.
Which elements have the highest electronegativities on the periodic table?
1 Answer
There isn't really a whole row that is the largest.
Explanation:
Elements from the halogen group including F, Cl, Br have pretty high electronegativities. The most electronegative element is Fluorine with a score of 4.0 (the highest possible.)
Across from Fluorine we also have N and O with high electronegativities.
Electronegativity is basically how much elements 'want' electrons. A simple way to think about it is that the closer an element is to Fluorine, the higher its electronegativity is.
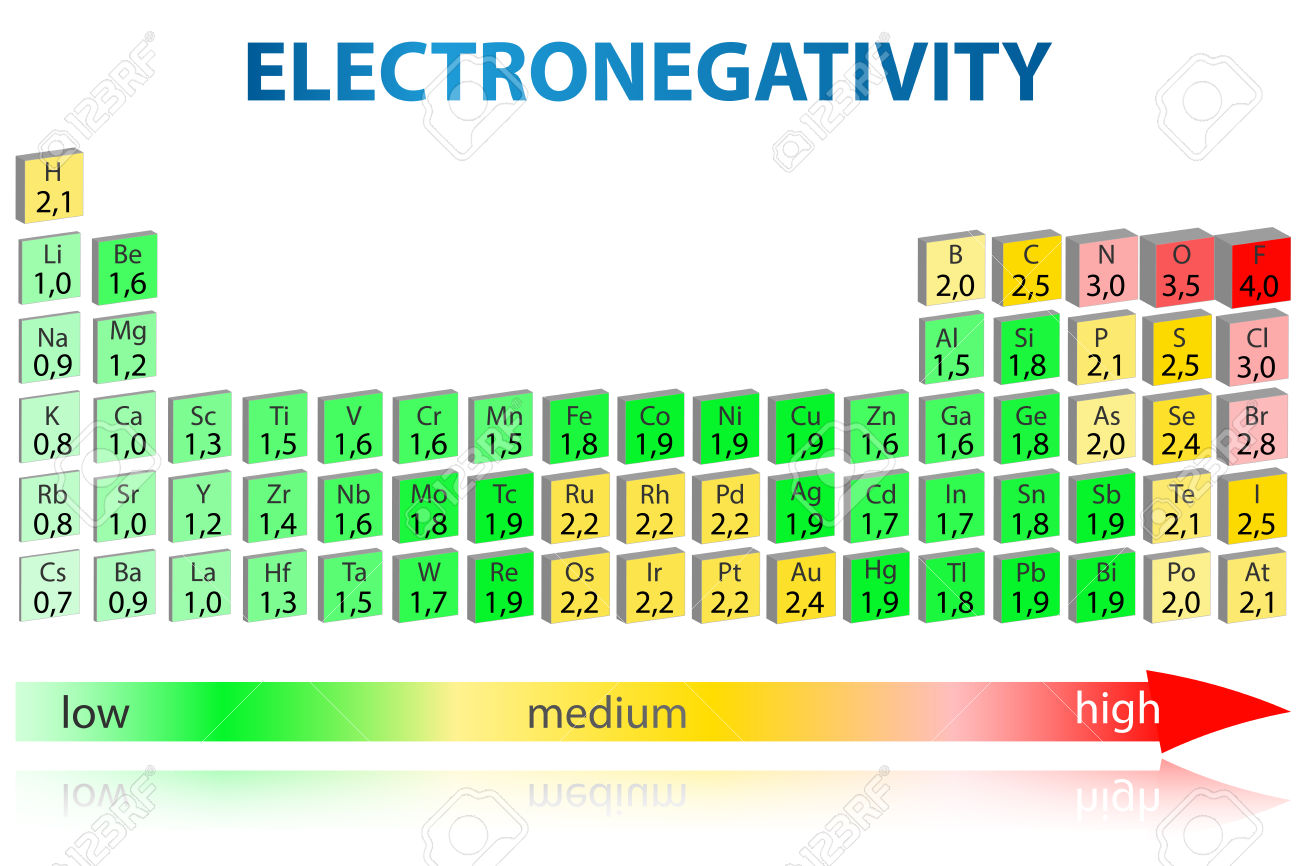
Electronegativity Elements Chart
Related topic
Electronegative Elements Tend To
Related questions
